Ai Ardlynoer*, Sussy Amogus*, Pipi Pampers*#
*Department of Chemical Biology, University of Massachusetts Cambridge, Jonestown, Guyana
#Division of Chess, Google, En Passant, Armenia
Modern molecular biology is often less of a science and more of an art. Though recent innovations have led to the development of powerful new techniques in genomics, proteomics, transcriptomics, epigenomics, lipidomics, lysozomics, PROTACSomics, hormonomics, mitrochondriomics, polysaccharidomics, and more, many of these methods rely on decades-old principles that present little novelty, particularly to the National Science Foundation Graduate Research Fellowships Program.1 This is most evident in our current methods of protein quantification. Perhaps the most common first step in any protein analysis project is analysis by SDS-PAGE gel. SDS-PAGE gels, though a staple of modern biochemistry and molecular biology, require lengthy setup time, rely on environmentally hazardous stains, and are highly tempting for budding PhD students to falsify.2 Chemical biology, the melding of organic synthesis with molecular biology, can offer a solution to this urgent problem. Here, we report Conrotatory Tandem Ring Inversion Verification Editing (ConTRIVEd) as a novel method of protein analysis.
ConTRIVEd allows for the label-free, high-throughput quantification of protein concentration via a click-capable EZ ligand, which can be easily synthesized in 32 steps. Treatment of sample with EZ, followed by analytical ultracentrifugation and mass spectrometry, easily separates proteins by molecular weight. Subsequent analysis with BYTE-Me software allows for quantification of molecular weight and easy identification of protein identity. ConTRIVEd therefore represents an exciting alternative to traditional biochemical methods.
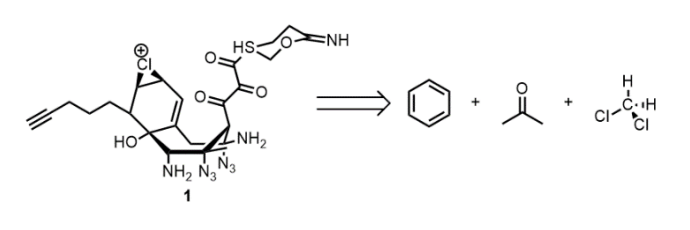
Synthesis of EZ
EZ ligand 1 was envisioned to be constructed from three fragments beginning with benzene, acetone, and dichloromethane respectively (Scheme 1). The synthesis of 1 is considered trivial and is left as an exercise to the reader.3 As a hint, in the final step, [4,12-diacetyloxy-15-(3-benzamido-2-hydroxy-3-phenylpropanoyl)oxy-1,9-dihydroxy-10,14,17,17-tetramethyl-11-oxo-6-oxatetracyclo[11.3.1.03,10.04,7] heptadec-13-en-2-yl] benzoate was stirred in a 1:1 mixture of 1,2-dioxane and water with 3 mol% Pd(PPh3)3 and heated at 235 °C overnight under N2. 1 was obtained as an off-white semi solid in 0.25% yield (100mg) over 32 steps. All compounds were purified by fractional distillation under slightly reduced pressure and characterized by 19F NMR spectroscopy.
ConTRIVEd Procedure
The azide and alkyne moieties on 1 allows for click chemistry of both azide and alkyne-labeled proteins. With our probe in hand, we set out to quantify the concentration of a model protein (Figure 1). Using amber codon suppression,4 an azidoalanine-substituted hemoglobin was expressed in Neisseria gonorrhoeae and purified by affinity chromatography. Purified protein was incubated with 1 and an ancient spell was cast over the mixture to promote copper-free alkyne-azide cycloaddition. The reaction mixture was subjected to analytical ultracentrifugation and the presence of hemoglobin in the sole fraction was confirmed by mass spectrometry and BYTE-Me software. The hemoglobin-1 complex was lyophilized and weighed to give the exact mass of protein in the original sample within 5%.

We therefore conclude that EZ ligand 1 successfully labeled hemoglobin, and ConTRIVEd successfully afforded the concentration of hemoglobin in vitro.
Conclusions
ConTRIVEd offers a fresh and exciting new approach to proteomics research and further development is worthy of a grant from the National Science Foundation. We fully expect ConTRIVEd to replace SDS-PAGE as standard procedure in molecular biology within 3 years. Interested parties may contact the authors at any time.
Conflicts of Interest
Ai Ardlynoer and Sussy Amogus declare their interest in this project to be more valid than Pipi Pampers’.
Acknowledgements
We acknowledge this paper happened.
Notes and references
1 Just trust me, bro
2 E. Bik, A. Casadevall, F.C. Fang, mBio. 2016, 7, 3, e00809-16
3 F. Professor, Soph. Orgo. 2018, 2, 6, 341-3654 P. Schultz, Magic Chem. 2001, 5, 2, 653-752
If you enjoyed this Immaterial Science article please like, share, and subscribe with your email, our twitter handle (@JABDE6), our facebook group here, or the Journal of Immaterial Science Subreddit for weekly content.
